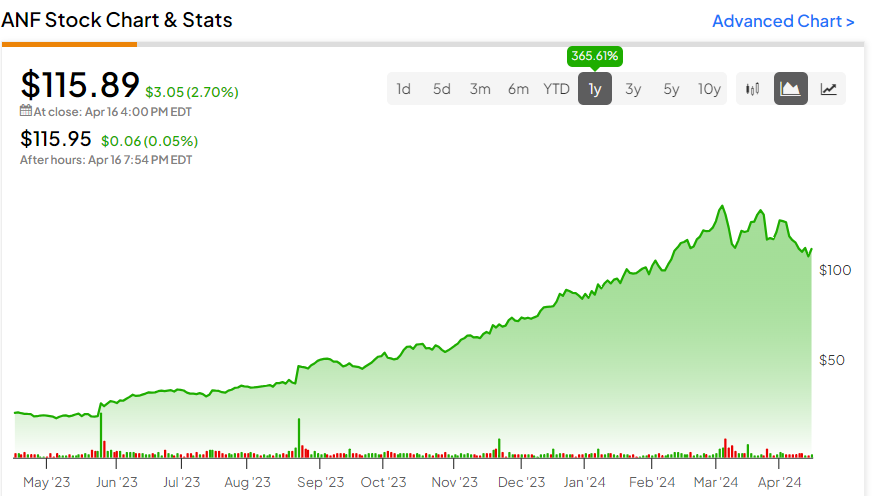
– On this exploratory substudy, therapy with acoramidis was related to attainable cardiac structural and practical enchancment in contrast with placebo, with potential cardiac amyloid regression
– The info display that concentrating on near-complete transthyretin (TTR) stabilization with acoramidis could allow cardiac reworking and practical restoration in sufferers with ATTR-CM
– These outcomes are the primary potential, longitudinal analysis of cardiac construction and performance by CMR imaging in a double-blind, placebo-controlled, interventional research in ATTR-CM
– The findings from this research construct upon optimistic outcomes from BridgeBio’s world ATTRibute-CM Part 3 trial, whereby the first endpoint was met (Win Ratio of 1.8) with a excessive statistical significance (p<0.0001); the substudy knowledge are in step with the cardiovascular medical advantages noticed with acoramidis
– Acoramidis was well-tolerated, with no security alerts of potential medical concern noticed
– BridgeBio’s New Drug Utility (NDA) has been accepted by the U.S. Meals and Drug Administration (FDA) with a PDUFA motion date of November 29, 2024; a Advertising Authorization Utility (MAA) for acoramidis has been accepted by the European Medicines Company (EMA), with an anticipated determination in 2025
PALO ALTO, Calif., April 07, 2024 (GLOBE NEWSWIRE) — BridgeBio Pharma, Inc. (Nasdaq: BBIO) (“BridgeBio” or the “Firm”), a commercial-stage biopharmaceutical firm centered on genetic illnesses and cancers, at the moment offered outcomes from the exploratory CMR imaging substudy of ATTRibute-CM, its Part 3 trial of acoramidis in ATTR-CM. These knowledge had been offered on the American Faculty of Cardiology (ACC) Annual Scientific Periods & Expo in a moderated poster session by Yousef Razvi, M.D. of College Faculty London. ATTRibute-CM was designed to check the efficacy and security of acoramidis, an investigational, next-generation, orally-administered, extremely potent, small molecule stabilizer of TTR. Based mostly on the optimistic outcomes from ATTRibute-CM, BridgeBio submitted an NDA to the U.S. FDA, which has been accepted with a PDUFA motion date of November 29, 2024, and an MAA to the European Medicines Company, with a call anticipated in 2025.
“CMR is the reference non-invasive imaging methodology to judge cardiac construction, operate, and amyloid burden in sufferers with ATTR-CM. The outcomes proven within the imaging substudy are indicative of potential enchancment of cardiac construction and performance in sufferers with ATTR-CM, in step with the medical outcomes with acoramidis therapy noticed within the ATTRibute-CM Part 3 medical trial. The influence on cardiac amyloid load highlights the potential that acoramidis might result in cardiac amyloid regression in a proportion of sufferers with ATTR-CM, which we now have not noticed in such a managed medical trial up to now,” mentioned Marianna Fontana, M.D., Ph.D., Professor of Cardiology and Honorary Advisor Heart specialist on the Nationwide Amyloidosis Centre, Division of Drugs, College Faculty London, and Principal Investigator of the Substudy.
CMR imaging was carried out at baseline earlier than the primary dose in 35 contributors or inside three months after the primary dose in 17 contributors (vary, 14-105 days); subsequent CMR imaging was carried out at months 12, 24, and 30 in these people who had been accessible to endure imaging, which was influenced by survival. Photographs had been learn centrally on the Nationwide Amyloidosis Centre in a style blinded to different medical knowledge. Findings included:
- Key CMR imaging parameters measuring cardiac construction, together with imply listed left ventricular mass, had been discovered to be secure or have a pattern in direction of enchancment on acoramidis vs. deteriorated on placebo over 30 months
- Therapy with acoramidis preserved or was related to a pattern in direction of enchancment in measures of cardiac operate together with left ventricular ejection fraction and stroke quantity in comparison with preliminary measures, and relative to placebo
In July 2023, BridgeBio announced optimistic outcomes from ATTRibute-CM, reporting a extremely statistically vital outcome, demonstrated by a Win Ratio of 1.8 (p<0.0001) on the first endpoint (a hierarchical evaluation prioritizing so as: all-cause mortality, then frequency of cardiovascular hospitalization, then change from baseline in N-terminal prohormone of mind natriuretic peptide, then change from baseline in 6-minute stroll distance). Acoramidis was well-tolerated, with no security alerts of potential medical concern recognized. BridgeBio has additionally offered analyses from ATTRibute-CM on the European Society of Cardiology Congress 2023 and on the American Heart Association Scientific Sessions 2023. In February 2024, BridgeBio shared optimistic outcomes of a single-arm Part 3 research of acoramidis in Japanese sufferers with ATTR-CM, together with no mortality noticed within the trial at 30 months.
About BridgeBio Pharma, Inc.
BridgeBio Pharma Inc. (BridgeBio) is a commercial-stage biopharmaceutical firm based to find, create, take a look at and ship transformative medicines to deal with sufferers that suffer from genetic illnesses and cancers with clear genetic drivers. BridgeBio’s pipeline of improvement applications ranges from early science to superior medical trials. BridgeBio was based in 2015 and its staff of skilled drug discoverers, builders, and innovators are dedicated to making use of advances in genetic medication to assist sufferers as rapidly as attainable. For extra info go to bridgebio.com and comply with us on LinkedIn and Twitter.
BridgeBio Pharma, Inc. Ahead-Trying Statements
This press launch accommodates forward-looking statements. Statements on this press launch could embrace statements that aren’t historic details and are thought of forward-looking throughout the that means of Part 27A of the Securities Act of 1933, as amended (the Securities Act), and Part 21E of the Securities Alternate Act of 1934, as amended (the Alternate Act), that are normally recognized by means of phrases resembling “anticipates,” “believes,” “estimates,” “expects,” “intends,” “could,” “plans,” “initiatives,” “seeks,” “ought to,” “proceed,” “will,” and variations of such phrases or related expressions. We intend these forward-looking statements to be lined by the protected harbor provisions for forward-looking statements contained in Part 27A of the Securities Act and Part 21E of the Alternate Act. These forward-looking statements, together with statements regarding the medical and therapeutic potential of our applications and product candidates, together with our medical improvement program for acoramidis for sufferers with transthyretin amyloid cardiomyopathy, the timing and success of our medical improvement applications, the progress of our ongoing and deliberate medical trials of acoramidis for sufferers with transthyretin amyloid cardiomyopathy, together with the expectations of receiving a PDUFA motion from the FDA on November 29, 2024 and receiving a call from the EMA on the MAA for acoramidis in 2025, the anticipated mental property safety of acoramidis, our deliberate interactions with regulatory authorities, the statements concerning the potential medical advantages or of potential advantages for ATTR-CM sufferers within the quotes of Dr. Fontana, and the timing of those occasions, replicate our present views about our plans, intentions, expectations and methods, that are primarily based on the data presently accessible to us and on assumptions we now have made. Though we imagine that our plans, intentions, expectations, and methods as mirrored in or steered by these forward-looking statements are affordable, we can provide no assurance that the plans, intentions, expectations, or methods will probably be attained or achieved. Moreover, precise outcomes could differ materially from these described within the forward-looking statements and will probably be affected by a lot of dangers, uncertainties and assumptions, together with, however not restricted to, preliminary and ongoing knowledge from our medical trials not being indicative of ultimate knowledge, the design and success of ongoing and deliberate medical trials, difficulties with enrollment in our medical trials, adversarial occasions that could be encountered in our medical trials, the FDA or different regulatory companies not agreeing with our regulatory approval methods, elements of our filings, resembling medical trial designs, conduct and methodologies, or the sufficiency of information submitted, potential adversarial impacts as a result of world COVID-19 pandemic resembling delays in regulatory evaluate, manufacturing and provide chain interruptions, adversarial results on healthcare techniques and disruption of the worldwide financial system, the impacts of present macroeconomic and geopolitical occasions, together with altering situations from the COVID-19 pandemic, hostilities within the Center East and Ukraine, growing charges of inflation and rising rates of interest, on our general enterprise operations and expectations, in addition to these dangers set forth within the Threat Elements part of our Annual Report on Type 10-Okay for the 12 months ended December 31, 2023 and our different filings with the U.S. Securities and Alternate Fee. Furthermore, we function in a really aggressive and quickly altering atmosphere through which new dangers emerge sometimes. These forward-looking statements are primarily based upon the present expectations and beliefs of our administration as of the date of this press launch, and are topic to sure dangers and uncertainties that might trigger precise outcomes to vary materially from these described within the forward-looking statements. Besides as required by relevant legislation, we assume no obligation to replace publicly any forward-looking statements, whether or not because of new info, future occasions or in any other case.
BridgeBio Media Contact:
Vikram Bali
contact@bridgebio.com
(650)-789-8220
The put up BridgeBio Pharma Presents Cardiac Magnetic Resonance (CMR) Imaging Proof In line with Scientific Enchancment Noticed within the ATTRibute-CM Part 3 Examine in Sufferers with Transthyretin Amyloid Cardiomyopathy (ATTR-CM) appeared first on FinanceGrabber.